The first time the new pancreas whispered to life inside a human body, the room went quiet. No dramatic beeping, no flashing lights, no cinematic countdown. Just a number on a monitor settling into a calm, steady rhythm, and a woman on the hospital bed feeling—if anything—nothing at all. Which, in a way, was the miracle. For the first time since she was a child, her blood sugar wasn’t a problem to solve. It had already been handled, invisibly, by something part human, part machine, and entirely new.
A Future That Clicks Into Place Under the Skin
Imagine waking up and not thinking about diabetes. No finger pricks, no carb math over breakfast, no quiet dread when the gym session runs long or traffic delays dinner. Instead, there’s a small, silent system pulsing away inside you—a cybernetic pancreas, watching, predicting, adjusting.
That’s the radical promise behind the world’s first functional “cyborg pancreas,” a bioelectronic organ that doesn’t just sit beside the body like a gadget, but lives within it, learns from it, and partners with it. It’s an invention that reads like science fiction and feels, to the people testing it, like getting their lives back in slow, astonished increments.
This isn’t simply a better insulin pump or a smarter sensor. It’s a living conversation between tissue and circuitry. Electrodes kiss the edges of nerves. Micro-reservoirs of insulin pulse like tiny mechanical heartbeats. Algorithms, trained on oceans of glucose data, sketch invisible curves into the next minutes and hours of your life. And bit by bit, the body—tired from years of being managed—starts to relax.
For decades, the holy grail in diabetes care has been something called the “artificial pancreas”: a closed-loop system that senses glucose and automatically adjusts insulin, with no human input. We’ve had pieces of that dream for a while—continuous glucose monitors, pumps, hybrid closed-loop systems—but they still orbit outside the body, clinging to skin with adhesives and infusion sets, asking for calibrations and confirmations and overrides. The cyborg pancreas slips beneath the surface and says: Let me handle this.
The Quiet War Inside a Diabetic Body
To understand how radical this is, you have to remember what diabetes really feels like—not the numbers and charts, but the body-level experience of living in constant negotiation with your own blood.
There’s the morning stomach flip when a meter blinks out a number that will decide how the day begins. The dry-mouth thirst that feels like you swallowed dust. The brain-fog slowness when sugar runs high, as though all your thoughts are dragging through syrup. Or the low—the dangerous low—that sweeps in quick and ghostly: shaky hands, tunnel vision, sweat like cold rain across your back as your brain quietly runs out of fuel.
This is what Type 1 diabetes is: an autoimmune ambush that destroyed the beta cells in the pancreas, those delicate islands of tissue that once released insulin in elegant, microscopic bursts. What the body once balanced unconsciously with exquisite timing, you now have to manage by hand, all day, every day, forever.
People often call diabetes a “numbers disease,” but that flattens the experience. It’s also a disease of anticipation. Every meal, every workout, every late-night snack comes with a guess: How will this hit my blood sugar? What’s the right trade-off between the risk of going too low and the damage of staying a little too high? Even with the best tech, there’s still a tax on your attention. You are the primary algorithm, constantly running.
The cyborg pancreas is built to relieve that burden—not only by correcting after the fact, but by predicting before trouble hits. It senses patterns—your patterns—and gradually shapes itself around them like a river finding its way around stones.
How a Cyborg Pancreas Learns to Be Human
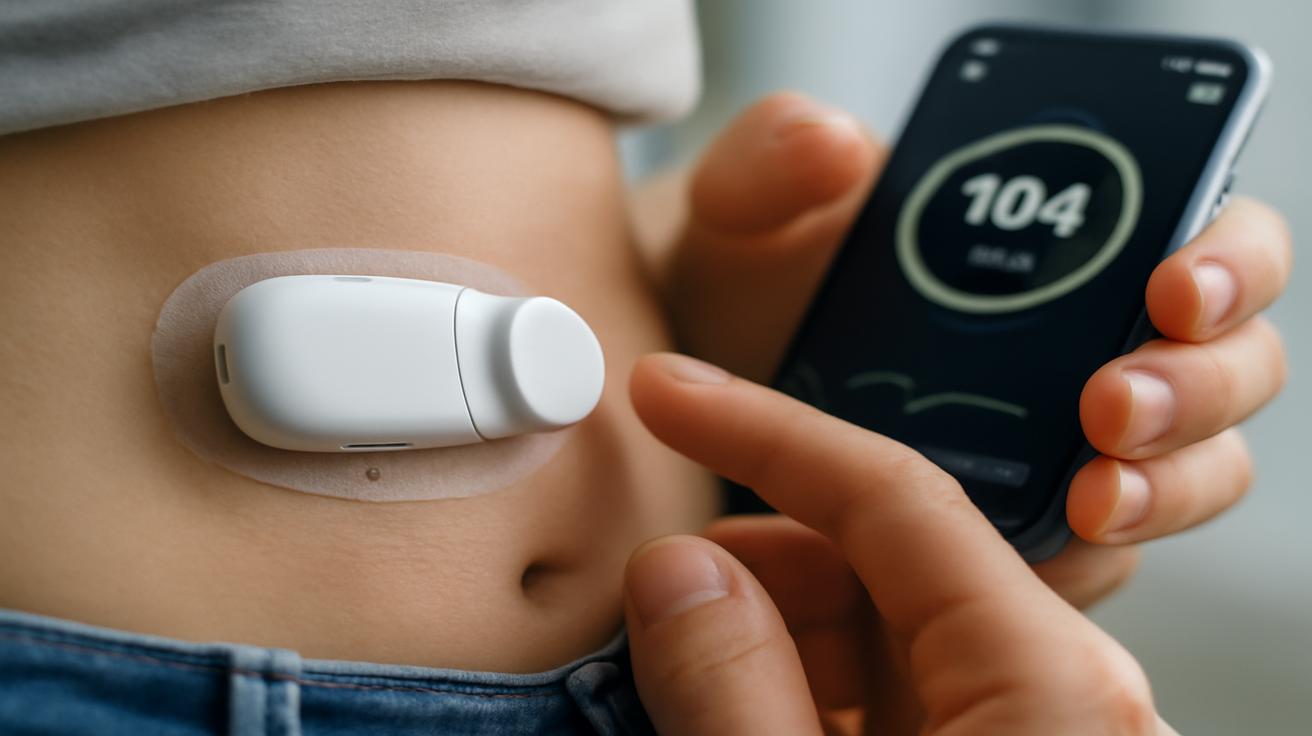
Despite the science-fiction name, the first cyborg pancreas isn’t some chrome-plated gadget glowing under the skin. It’s surprisingly un-cinematic: a compact, biocompatible housing, the color of bone, nestled near abdominal tissue. Inside, there’s a small reservoir of insulin, a set of ultra-fine electrodes, a glucose-sensing array, and a tiny powerhouse of processors and wireless coils.
The magic lies in how it listens.
Instead of just reading glucose from the fluid beneath your skin like traditional sensors, this device eavesdrops on the physiological chatter around it—subtle electrical changes, temperature shifts, micro-movements in blood flow. It builds a picture of your metabolic rhythm. Early versions are paired with an external charging pad and a backup monitor, but the real work happens inside, beneath the feel of your fingers.
At the core is an adaptive algorithm, trained on millions of hours of glucose and insulin data from people all over the world. When a surgeon implants the device, that algorithm doesn’t start from zero—it begins with a robust sense of “typical” diabetic physiology. Then, over the next days and weeks, it starts to unlearn the average and learn you.
It grows familiar with how your blood sugar responds to stress, or missed meals, or late-night snacking. It picks up on the little seasonal shifts: how winter inactivity nudges your numbers higher, how summer heat changes your insulin sensitivity. At first, doctors watch closely, checking data daily. But soon those checks stretch to weeks, then months. The device begins to make decisions that feel uncannily like what you might have done yourself—only sooner, smoother, and without waking you at 3 a.m.
It’s not just “automatic insulin delivery”; it’s more like metabolic companionship. Your pancreas once knew you like this. Now, in a cyborg twist, something else is learning the same intimate language.
| Feature | Conventional Diabetes Tech | Cyborg Pancreas |
|---|---|---|
| Location | Outside the body (wearables) | Implanted, fully internal |
| User Input | Frequent: boluses, calibrations, mode changes | Minimal: occasional check-ins, rare overrides |
| Sensing | Interstitial glucose only | Glucose + neural, thermal, and flow signals |
| Algorithm | Preset rules, limited personalization | Adaptive, continuously learning model |
| Daily Burden | Constant attention, visible devices | Mostly invisible, low mental load |
Between Flesh and Circuit: What Makes It “Cyborg”
The word “cyborg” still pulls our imagination toward science fiction—glinting eyes, titanium limbs, people more machine than human. But in its simplest form, a cyborg is just a being whose organic functions are permanently entwined with technology. Pacemakers made our hearts a little cyborg decades ago. Cochlear implants did the same with hearing.
This new pancreas carries that idea deeper, into the quiet housekeeping of metabolism. Its creators didn’t set out to make people robotic; they wanted to make them less preoccupied with the machinery of their illness. The technology is cyborg not because it replaces humanity, but because it restores something that disease took away.
What makes this system feel qualitatively different from a pump-and-sensor setup isn’t just where it sits. It’s how the boundaries blur. Electrodes brush nerve fibers that once listened to real pancreatic activity. Signals pass both ways. In early lab animals, those connections began as simple stimulus and response; over time, though, the nervous system rewired around the artificial organ, treating it less like a foreign object and more like a strangely-behaving part of itself.
In humans, early participants describe an eerie shift: a sense that their energy is steadier, their moods less yanked around by glucose swings. One man in his thirties compared it to “finally driving on cruise control after years of riding the brakes.” Another noticed that the background hum of anxiety he had lived with since childhood “went quiet in a way I didn’t know was possible.”
There is a psychological cyborg moment, too—the first time you eat a meal and don’t mentally calculate the insulin. The first time you go to sleep without setting a 2 a.m. alarm to “just check.” The first time you realize you’ve gone half a day without thinking about your numbers, and that this is not neglect, but trust.
The Radical Relief of Invisible Care
This is where the revolution truly lives: not in the chip specifications, but in the erosion of fear. Diabetes has always been both a medical condition and a lifestyle occupation. Even people with the best tools still carry constant vigilance: is my sensor adhesive peeling, did my pump site kink, did I pack enough supplies just in case?
With an implanted system, the visibility shrinks. There’s nothing taped to your arm at the beach, no tubing to catch on doorknobs, no box buzzing conspicuously on your waistband at a dinner table. For children and teenagers, that invisibility carries a special kind of grace—they get to be regular kids in bodies that are quietly, cleverly augmented.
But invisibility has a shadow side: loss of control can feel frightening. For years, person with diabetes have been the pilots of their own survival; handing the controls to an algorithm takes a leap of faith. That’s why early trials build in layers of reassurance: backup fail-safes, override options, a visible data stream for those who want to keep watching. The goal is not to erase the person from the loop, but to make their role optional rather than compulsory.
Who Gets a Cyborg Organ, and Who Gets Left Behind?
As soon as you talk about a groundbreaking device like this, another question stands at the door: Who will get it first, and who might never get it at all?
Early cyborg pancreas implants have gone, predictably, to a small group of volunteers: people with severe, brittle Type 1 diabetes, frequent dangerous lows, or cases where existing tech simply couldn’t keep up. They tend to live near major research hospitals. They have access, time, and resources. It’s a familiar pattern in medical innovation: the frontier is narrow and privileged at first.
The teams behind this technology know that pattern, and some are already talking about tiered versions of the device—simpler, cheaper models for health systems in low-resource settings, modular upgrades, flexible programming that can adapt to different insulin formulations. But there’s no escaping a hard truth: complex implantable biotech is expensive, and diabetes is a global disease that disproportionately affects people with the least access to cutting-edge care.
This is where policy, ethics, and engineering have to move in concert. Insurance frameworks will need to recognize not just the gadget itself, but the lifetime savings in reduced complications: fewer ER visits for lows, fewer kidney failures and amputations and blindness decades later. Governments will need to negotiate pricing and incentivize equitable rollout. Clinicians will need training to implant and manage these devices safely, not just in elite centers but in ordinary hospitals.
If the cyborg pancreas is to be more than a curiosity for the few, its story must bend toward scale and justice. Otherwise, it risks becoming a symbol of what biotech can do for those already standing near the top of the care ladder.
The Body’s Immune System, and Other Complications
Then there’s the body itself, which has strong opinions about things left inside it.
Any implanted device has to survive a long, suspicious stare from the immune system. Proteins coat its surface. Cells probe, then attack. Scar tissue tries to wall it off like a stubborn landlord. If the cyborg pancreas is going to last for years, it needs clever disguises: biocompatible coatings, microtextures that discourage inflammation, designs that remain accessible to blood and interstitial fluid without triggering alarm bells.
And what about failure? No device lasts forever. Batteries degrade, reservoirs empty, components age. The designers have built in ways to sense wear and tear and send quiet alerts long before things go wrong. In some configurations, the external charger also serves as a diagnostic wand, reading deeper than a smartwatch ever could. Removal and replacement become surgical, yes, but increasingly routine—like swapping out an old pacemaker for a new one.
These are the unglamorous logistics of cyborg life: follow-up appointments, firmware updates, the slow, careful dance of medicine and engineering keeping pace with each other inside your abdomen.
Living With Tomorrow Inside You
What does it feel like to be one of the first humans walking around with a cyborg pancreas? The answers are still accumulating, but they share a certain quiet awe.
Participants in early trials talk less about the drama of surgery and more about the strange ordinariness that follows. The first grocery run where the dessert aisle no longer feels like a minefield. The first hike without stuffing their bag with juice boxes and glucose tabs. The realization, a few months in, that their HbA1c—the long-term measure of glucose control—has drifted into near-normal ranges without crash diets or hypervigilance.
Some struggle, at first, with a sense of identity. For years, diabetes was not only a condition but a community, a badge of resilience. To hand much of that labor to a device can feel like losing a part of yourself, as if a finely honed survival skill has been retired. But over time, that feeling seems to soften into something else: an acceptance that being tough was never the point. Being free was.
There’s also a more subtle mental shift: trusting your own body again. After years of treating it like an unreliable narrator—always second-guessing hunger, thirst, fatigue, wondering if a mood is real or just blood sugar—people begin to listen with less suspicion. If they’re tired, maybe they just need rest, not a meter. If they cry, maybe it truly is about the movie, not the numbers.
The cyborg pancreas doesn’t cure diabetes. The underlying autoimmune storm is still there. But it changes the daily choreography so completely that the disease steps, for many hours at a time, out of the foreground and into the wings.
The Radical Path Ahead
It’s tempting to think of this first cyborg pancreas as an endpoint—a long-awaited finish line in the marathon of diabetes research. In reality, it’s more like a gate swinging open onto a much stranger landscape.
Once we accept that organs can be partially replaced by integrated machines, new questions rise fast. Could similar systems help people with Type 2 diabetes whose pancreases still sputter but no longer coordinate well? Could adaptive implants someday share data with the liver, the gut, even the brain, weaving a fuller metabolic network? Might we eventually build hybrid organs that not only mimic lost function but enhance it, edging into territory that looks less like therapy and more like upgrade?
With each step comes an ethical tangle. How much of our inner workings are we willing to outsource to algorithms? Who owns the intimate data pouring out of an internal device—the patient, the hospital, the company that made it? How do we ensure that “better” does not become compulsory, that people can say no to internal tech without being left behind?
For now, though, the story remains anchored in something more humble and more human. It is the story of a woman waking up in a hospital bed and not feeling much of anything at all, because for the first time since childhood, her body’s most basic balancing act is quietly, competently handled. It is the story of a small device humming along where a biological organ once flourished, not flashy, not heroic, just steady.
Between the rhythms of pulse and breath, another rhythm has joined: the minute-by-minute rise and fall of glucose, traced and steadied by a silicon partner that never sleeps. In that partnership—messy, imperfect, astonishing—lies a new way of thinking about illness, technology, and what it means to live in a body that is both fragile and endlessly modifiable.
The first cyborg pancreas is more than a medical milestone. It is a quiet, radical invitation to imagine a world where diabetes no longer commands every thought, where the hardest work of survival can be gently handed off, and where the line between flesh and circuit becomes not a wound, but a seam.
Frequently Asked Questions
Does a cyborg pancreas cure diabetes?
No. The underlying autoimmune or metabolic causes of diabetes remain. The cyborg pancreas manages blood sugar so effectively that many daily symptoms and risks are reduced, but it does not restore natural pancreatic cells or stop the disease process.
How is an implanted pancreas different from current insulin pumps?
Current pumps and sensors sit outside the body and require frequent user input and maintenance. A cyborg pancreas is implanted inside the body, senses multiple physiological signals, and uses adaptive algorithms to make most decisions automatically, with far less daily involvement from the user.
Is it safe to have electronics permanently inside the body?
Implantable devices like pacemakers and neurostimulators have shown that carefully designed electronics can function safely for years inside the body. The cyborg pancreas uses biocompatible materials and multiple safety layers, but long-term data is still being collected as trials continue.
Will everyone with diabetes eventually be able to get a cyborg pancreas?
Access will depend on regulatory approval, cost, healthcare coverage, and local medical infrastructure. The technology is promising, but widespread availability—especially in low-resource settings—will take time and deliberate policy work to achieve.
Can the user still control or override the device?
Yes. Early versions include options to view data, adjust certain settings, and override automated decisions when necessary. The aim is to reduce daily burden, not remove agency. As the system proves itself, many users choose to intervene less often, but the option to step in remains.